Current Pediatric Research
International Journal of Pediatrics
Case Report of Complex Congenital Heart Block in a Term Female Nigerian Neonate.
Uchechukwu Chukwuebuka1*, Chinwe Dorothy Obu1, Ukoh Fortune Amuche Ujunwa2, Obumneme Benaiah Ezeanosike1, Ikenna Chukwudebelu Nwokoye1
1Department of Pediatrics, University of Alex Ekwueme Federal Teaching Hospital, Abakaliki, Nigeria
2Department of Pediatrics, University of Nigeria, Enugu, Nigeria
- Corresponding Author:
- Uchechukwu Chukwuebuka
Department of Pediatrics,
University of Alex Ekwueme Federal Teaching Hospital,
Abakaliki,
Nigeria,
E-mail: ukohuc@yahoo.com
Received: 29 May, 2024, Manuscript No. AAJCP-21-41586; Editor assigned: 03 June, 2024, Pre QC No. AAJCP-21-41586 (PQ); Reviewed: 17 June, 2024, QC No. AAJCP-21-41586; Revised: 21 June, 2024, Manuscript No. AAJCP-21-41586 (R); Published: 28 June, 2024, DOI:10.35841/0971-9032.28.06.2270-2272.
Congenital heart block (CHB) is a rare disorder that has high morbidity and mortality especially when associated with structural congenital heart defects and is usually undiagnosed. It is important to have a high index of suspicion for early diagnosis and as well as an aggressive therapeutic approach when appropriate. We report a case of a term, small-for-gestational-age female neonate who had complete congenital heart block associated with congenital heart defect complicated by congestive cardiac failure. This highlights challenges in early diagnosis and prompt management of complex CHB in a low resource setting and the importance of quality antenatal care and proper screening for early antenatal diagnosis and planned perinatal and early postnatal management when appropriate in order to optimize outcome.
Keywords
Congenital heart block, Congenital heart defect, Term neonate, Antenatal.
Introduction
Congenital Heart Block (CHB) is a rare disorder with an estimated incidence of about 1 in 22,000 live births. CHB occurs due to impairment in the conduction system causing delay or block in the transmission of atrial impulse to ventricles [1]. Two forms of CHB exist namely isolated CHB and complex CHB. Isolated CHB is defined as congenital heart block found in a structurally normal heart and occurs due to the presence of maternal auto-antibodies, anti-Sjogren’s- Syndrome-related antigen A autoantibodies (anti-Ro/SSA) and anti-Sjogren’s-Syndrome-related antigen B autoantibodies (anti-La/SSB), that are transferred across the placenta to the fetus damaging the fetal heart conducting system. Complex CHB occurs when it is present with congenital structural abnormality of the heart [2].
It is important to distinguish these two forms of CHB because they differ not only in their pathogenesis and in their rate of recurrence in subsequent siblings, but also in the prognoses of affected children. Infants with CHB associated with severe structural heart disease have a poorer prognosis than infants with isolated CHB, while the risk of recurrence is higher in mothers who have tested positive for anti-Ro/SSA antibodies [3]. Although it is an uncommon disorder, CHB may be associated with a high morbidity and mortality. As a result, it requires a high index of suspicion when there is a finding of fetal bradycardia for early diagnosis and appropriate planning of perinatal management in centers with facilities for pacemaker treatment, but this is sometimes not possible in low resource settings [4].
Case Presentation
A female neonate who was delivered through Emergency Lower Segment Caesarean Section (EmLSCS) at gestational age of 38 weeks to a 24 year para4+1A3. The indication for EmLSCS was fetal bradycardia diagnosed by ultrasonography done at onset of labour. Ultrasonography was indicated by fetal bradycardia noted with the fetoscope in a peripheral hospital. Mother had no history of rash or febrile illness before or during pregnancy. She received only routine antenatal medications during pregnancy [5]. There was neither history of congenital heart disease in the family nor any history of recurrent pregnancy loss. Membranes were ruptured during labour which was not prolonged. Baby was delivered with low Apgar score (21, 35 and 510) and established spontaneous respiration after resuscitation with intermittent positive pressure ventilation and chest compression for a period of 20 mins. Thereafter, baby remained clinically stable apart from persisting low heart rate of 55 bpm-60 bpm [6].
Examination findings after resuscitation in neonatal ward showed a small for gestational age newborn with head circumference of 32 cm, length 45 cm and weight 2.2 kg. She was conscious and had widely patent anterior fontanelle and the tone was normal in all limbs. The heart rate remained at 55 bpm-60 bpm, apex beat was located at 4th left intercostal space in the mid-clavicular line, with grade 3/6 pansystolic murmur maximal at the left lower sternal border.
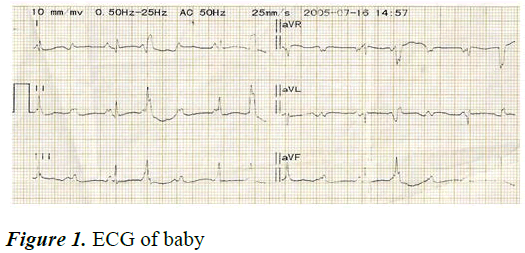
There was hepatomegaly. Blood pressure was not done. The peripheral capillary oxygen saturation (SpO2) measured at different times ranged between 87%-93%. Other systems were essentially normal [7]. Her investigation results showed Packed Cell Volume (PCV) of 37%, leucocyte total and differential counts, blood glucose level, serum sodium, potassium, calcium, urea and creatinine levels were within normal range. Twelve lead Electrocardiogram (ECG) showed bradycardia, P-mitrale, Atrio-Ventricular (AV) dissociation, left axis deviation, widened QRS complex and ST segment depression (Figure 1). A clinical diagnosis of atrioventricular canal defect in a child with complete congenital heart block in background symmetrical intrauterine growth restriction was made. Routine neonatal care was provided for her in addition to antibiotic therapy [8]. On the 4th day of life, baby developed periorbital puffiness, bilateral pitting pedal oedema, abdominal distension and increased respiratory rate (80 c/m) and pallor; heart rate was noted to be 77 bpm. Intravenous frusemide was added. She died on the 5th day of life [9].
Results and Discussion
CHB carries a significant risk of morbidity and mortality (15%-30%), especially in utero or in the first few months of life. About 63% of all recognized cases are reported to require pacemaker implantation before reaching adulthood [10].
Congenital heart block can be detected in utero between 18 and 28 weeks of gestation by foetal echocardiography in the presence of fetal bradycardia. In utero diagnosis of congenital heart block is associated with structural heart disease in approximately one half of the cases. Few cases of CHB associated with structural heart defects have been reported in Sub-Saharan Africa (SSA). One of the first cases of CHB in Africa was reported by Okoromaet, al., in Nigeria in a 3- month-old infant who was successfully treated with pacemaker implantation.
Our patient was diagnosed late through ultrasonography performed at term for fetal bradycardia while mother was in labour. Baby had a heart rate of 50-60 beats/min at birth whereas normal newborns have a heart rate of 120-160 beats/ min. The ECG showed the most severe type of heart block, third degree heart block, where there is no transmission of atrial impulse to the ventricle (Figure 1). In less than 10% of cases, CHB may be either 1st or 2nd degree block at birth, of which 50% progress to 3rd-degree block in postnatal life. Third-degree heart block is diagnosed when there is no association between P waves and QRS complexes as was seen in our patient.
Two thirds of all CHB patients require pacemaker implantation and it is recommended in symptomatic bradycardia. Third degree CHB is typically irreversible, has a mortality rate of 30% and requires pacemaker placement in up to 66% of cases. 8 It is also recommended in infants with CCHB and heart rate less than 55 beats/min in the absence of structural heart disease or with rate less than 70 beats/min with concomitant congenital heart disease or signs of ventricular dysfunction. There is a higher association of congenital heart block occurring with congestive heart failure in utero, and thus a poorer prognosis. Our index patient did not have features of congestive heart failure at birth but developed congestive heart failure by 4th day of life leading to early demise.
The outcome for patients with congenital heart block depends largely on the presence or absence of underlying structural heart disease, a heart rate less than a critical value, frequently quoted as 55 bpm and the presence or absence of congestive heart failure. If the heart block is diagnosed as a bradycardia during the fetal period, there is a very high rate of fetal and neonatal loss. These were found in the index patient who had pansystolic murmur, fixed splitting of second heart sound, low heart rate and developed congestive cardiac failure leading to her demise in the first week of neonatal life.
Complex CHB has a higher morbidity and mortality than isolated CHB and this is determined more by the underlying structural congenital heart disease than by the need for a pacemaker per se. Mortality is highest in the neonatal period and association with major cardiac malformations is a bad prognostic sign. In utero treatment and referral to a specialized cardiac centre was not done in this patient because of late presentation to our hospital as this may have improved outcome.
Conclusion
This report highlights the importance of early detection of complex CHB through quality antenatal care as well as timely initiation of management of such neonates for better outcome. In resource poor countries like ours, early thorough physical examination and ultrasonography can be diagnostic in mothers presenting with fetal bradycardia. The delivery of such babies should be planned in a tertiary care center where pacemaker placement facility and other necessary cardiac interventions are available.
References
- Dey M, Jose T, Shrivastava A, et al. Complete congenital foetal heart block: A case report. Facts Views Vis Obgyn 2014; 6(1): 39-42.
[Google Scholar] [PubMed]
- Rosenthal E. Classification of congenital complete heart block: Autoantibody-associated or isolated? Lupus 2003; 12(6): 425-426.
[Crossref] [Google Scholar] [PubMed]
- Friedman D, Lj D, Glickstein J, et al. A review of congenital heart block. Images Paediatr Cardiol 2003 1; 5(3): 36-48.
[Google Scholar] [PubMed]
- Okoroma EO, AGHAJI MC. Congenital complete heart block: Treatment by pacemaker implantation in a 3 month old Nigerian child. Cardiol Trop 1987; 13(52): 167-170.
- Jaeggi ET, Hamilton RM, Silverman ED, et al. Outcome of children with fetal, neonatal or childhood diagnosis of isolated congenital atrioventricular block: A single institution’s experience of 30 years J Am Coll Cardiol 2002; 39(1): 130-137.
[Crossref] [Google Scholar] [PubMed]
- Buyon JP, Hiebert R, Copel J, et al. Autoimmune-associated congenital heart block: Demographics, mortality, morbidity and recurrence rates obtained from a national neonatal lupus registry. J Am Coll Cardiol 1998; 31(7): 1658-1666.
[Crossref] [Google Scholar] [PubMed]
- Karnabi E, Boutjdir M. Role of calcium channels in congenital heart block. Scand J Immunol 2010; 72(3): 226-234.
[Crossref] [Google Scholar] [Pubmed]
- Yildirim A, Sedef Tunaodlu F, Karaadac AT. Neonatal congenital heart block. Indian Pediatr 2013; 5(50): 483-488.
[Crossref] [Google Scholar] [PubMed]
- Kiblawi MA, Naeem A, Al Attrash EA, et al. Complete congenital heart block in a newborn associated with maternal systemic lupus erythematosus: A case report. Intl J Med Stud 2013; 1(30): 128-131.
- Yan J, Varma SK, Malhotra A, et al. Congenital complete heart block: Single tertiary centre experience. Heart Lung Circ 2012; 21(11): 666-670.
[Crossref] [Google Scholar] [PubMed]